|
1. Combine 1 volume of cell suspension and 1 volume of trypan blue (0.4%).
For example, 50 ml of cells and 50 ml
of trypan blue. Mix thoroughly and allow to stand for 5 minutes.
2. Rinse the hemocytometer with water, and then ethonal (70%) and wipe clean with kimwipes.
3. With the cover slip in place, transfer a small amount of trypan blue-cell
suspension to both chambers of the hemocytometer by carefully touching
the edge of the cover slip with the pipette tip and allowing each chamber
to fill by capillary action.
4. Count all cells in the 1mm center square and four 1 mm corner
squares. Viable cells don't take up trypan dyes, wherease dead cell do.
Keep a separate count for viable and non-viable cell.
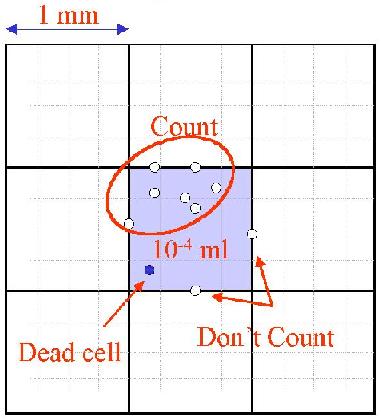
5. Count cells on top and left touching middle line. Do not count cells
touching middle line at bottom and right.
6. The total number of cell count must be greater than 100. Otherwise count more squares.
7. Each 1 mm square represents a total volume of 0.1 mm3
or 10-4 ml.
Viable cell count (cells/ml) = average cell count per 1mm square
* 2 * 104
8. Clean the hemocytometer with water.
|